-Delile
Ai-oxide iyinhlanganisela yamakhemikhali evela ezinhlanganisweni ze isici sensimbi noma okungeyona insimbi ne-oxygen. Ekubunjweni kwamakhemikhali, i-reagent (i-metal + oxygen) ithathwa ohlangothini lwesobunxele futhi umkhiqizo wakhiwa kuyo ohlangothini lwesokunene. Isibonelo, inhlanganisela ye-calcium ne-oxygen izokhiqiza ngqo i-calcium oxide.
Eqinisweni, ngokuvamile oxides Akhiwa ezimweni lapho izakhi zamakhemikhali zihlangana nomoya noma amanzi, anobukhona obukhulu bomoya-mpilo: lokhu kubangela ukugqoka ezintweni, ikakhulukazi uma kukhulunywa izinsimbi. Ukulungisa lokhu, izinto ze-antioxidant zivame ukusetshenziswa.
Ngaphakathi kwama-oxide, ukuhlukaniswa kuvame ukwenziwa ngokuya ngento i-oxygen ehlanganiswe nayo:
- Ama-oxide ayisisekeloUmkhiqizo ohlangene wokuhlanganiswa kwento yensimbi ne-oxygen.
- Ama-oxide e-acidUmkhiqizo oyinhlanganisela wenhlanganisela yento engeyona eyokulinganisa enomoya-mpilo.
- I-oxide ye-Amphoteric: i-amphoteric element ibandakanyekile ku-compound, ngakho-ke ama-oxides asebenza njengama-acids noma izisekelo.
Isibongo
Ukuqamba lezi zinhlobo zezinto, kunezindlela ezintathu zokukwenza:
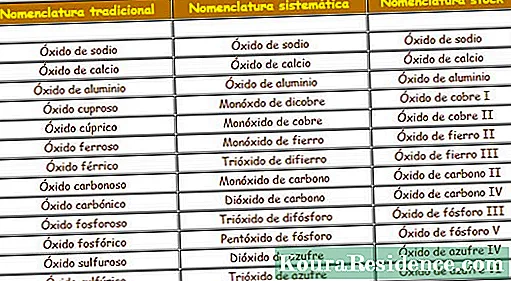
I- isibizo sendabuko (noma i-stoichiometric): Yiyona eqamba igama i-valence yesici esithile segama, ngochungechunge lweziqalo nezijobelelo. Indlela i-oxide ngayinye ebizwa ngayo iyahlukahluka ngokuya ngenani lama-valence isici esinawo.
- Lapho i-elementi ine-valence eyodwa kuphela, i-oxide izobizwa ngokuthi 'i-oxide (futhi into enesijobelelo esakhelwe ngaphakathi' ico ', njenge i-potassium oxide)’
- Lapho i-elementi inama-valence amabili, i-oxide izobizwa ngokuthi 'i-oxide (futhi into enesijobelelo esakhelwe ngaphakathi' ico ', njenge i-ferric oxide) 'Okwe-valence enkulu kakhulu, ne-'xide (kanye ne-elementi enesijobelelo esakhelwe ngaphakathi' ibhere ', njenge i-oxide eyisiqhuma)’
- Lapho i-elementi inama-valence amathathu, i-oxide izobizwa ngokuthi 'oxide (futhi into enesiqalo' hiccup 'kanye nesijobelelo' bear ', njenge i-oxide ye-hyposulfurousKu-valence ephansi kunazo zonke, izobizwa ngokuthi 'i-oxide (futhi into enesijobelelo' ibhere ', njenge i-oxide enesibabule) kwe-valence emaphakathi, kanye ne-'xide (kanye nento enesijobelelo esishumekiwe 'ico', njenge i-sulfuric oxide)’
- Lapho i-elementi ine-valence ezine, i-oxide izobizwa ngokuthi:
- 'I-oxide (kanye ne-elementi enesiqalo esithi' hiccup 'kanye nesijobelelo' bear ')' se-valence ephansi kakhulu. Ngokwesibonelo, i-oxidehypochlorous.
- ‘I-oxide (kanye ne-elementi enesijobelelo‘ ibhere ’) le-valence yesibili encane kunazo zonke. Ngokwesibonelo, i-chloride oxide.
- 'I-oxide (kanye ne-elementi enesijobelelo esakhelwe ngaphakathi' '') 'se-valence yesibili ngobukhulu. Ngokwesibonelo, i-chloride oxide.
- 'I-oxide (kanye ne-elementi enesiqalo' ngakunye 'nesijobelelo' '') se-valence enkulu kakhulu. Ngokwesibonelo, i-perchloric oxide.
I- isibizo esihleliwe Ilula kunaleyo yendabuko, futhi i-oxide ne-element ziyabizwa, kepha ngaphambi kokuba kubhalwe inani ngalinye lama-athomu anawo kulelo molecule. Isiqalo 'mono' kuzoba nge-athomu elilodwa, isiqalo 'di' kwababili, u-'tri 'kwabathathu,' i-tetra 'yabane, i-'penta' yabahlanu, i-'hexa 'eyisithupha, i' i-hepta 'kwabayisikhombisa ne-'octo' kwabayisishiyagalombili. Leli qembu lifaka, isibonelo, i- i-dichopper monoxide, i-dialuminium trioxide, isikhutha, noma i-difluorine monoxide.
I- Igama lesitokoEkugcineni, kusekelwe ekubhaleni igama elithi oxide, elilandelwa igama lensimbi kanye nenombolo ye-oxidation noma ye-valence esebenza ngayo, kubakaki nakubanombolo bamaRoma. Ngokufana negama lesibizo lendabuko, kuzobhalwa i-chlorine (I) i-oxide i-hypochlorous oxide, i-chlorine (II) i-oxide i-chloride oxide, i-chlorine (III) i-oxide i-chloric oxide, futhi i-chlorine (IV) i-oxide i-perchloric oxide.
Landela nge:
- Ngabe i-acids ibizwa kanjani?


